Crystal Structure Prediction Services
Find stable polymorph, fast.
Experimental methods alone cannot explore all stable crystal forms of a drug. Computational predictions can assist in identifying the potential risks of polymorphism and in finding crystal forms that are better suited for formulation with respect to physico-chemical properties.
OpenEye offers custom Crystal Structure Prediction (CSP) for drug-like molecules with a fast turnaround time, exhaustively sampling all crystal packings, and providing accurate yet cost-effective solutions for your drug development needs.
Features
-
Fast Turnaround Time
-
Requires only 2D Structures of the Active Pharmaceutical Ingredient (API)
-
Utilizes Custom Crystal Force Fields and Quantum Mechanics
-
Identify Polymorphs at Room Temperature
-
Accurate and Validated against Diverse Molecules

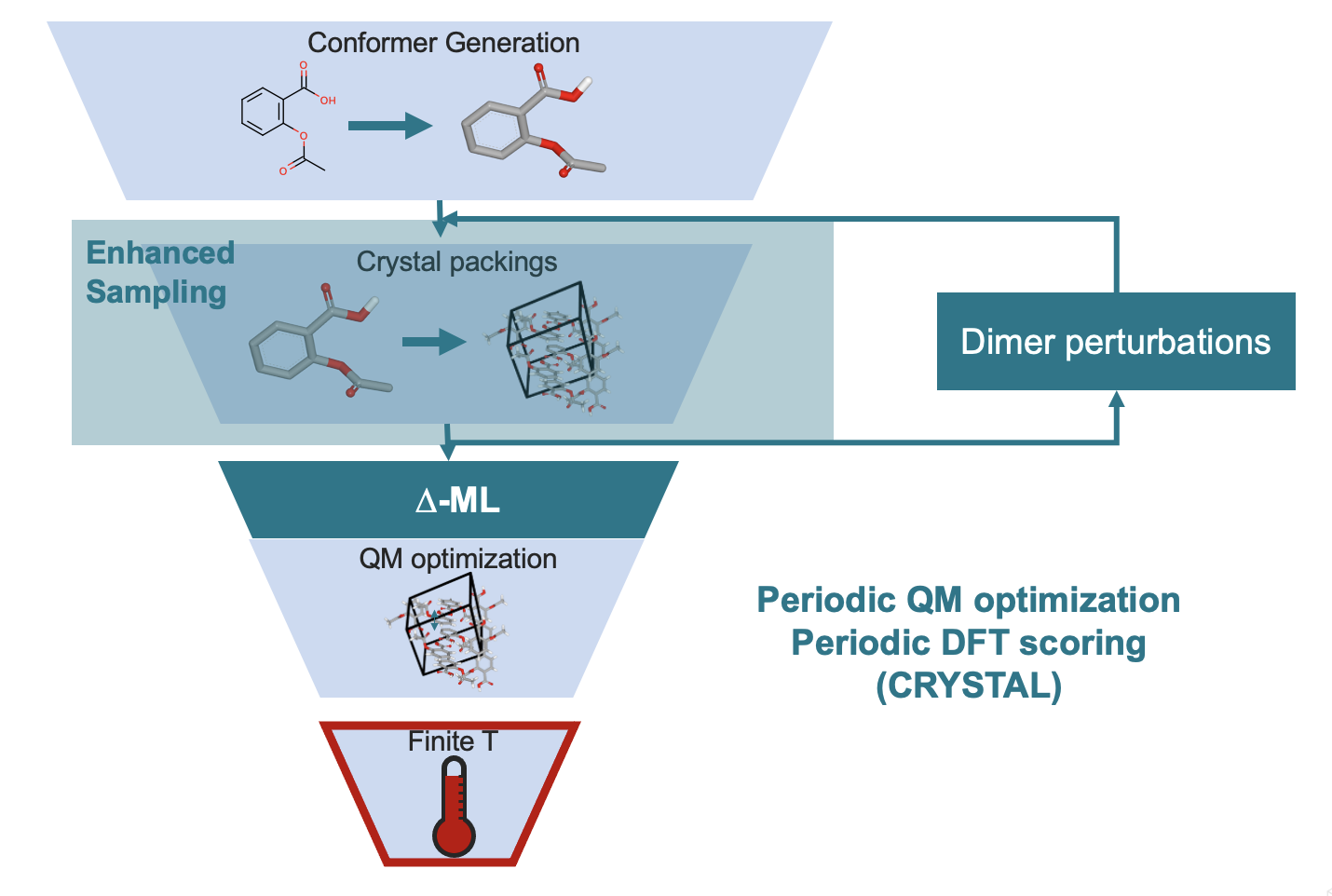
Crystal structure prediction (CSP) of drug-like molecules presents several challenges, such as unknown tautomer states, novel chemical motifs, high flexibility, intricate hydrogen bonding networks, among others.
Traditional approaches to CSP for drug molecules can take weeks to months. A significant bottleneck in these methods involves periodic QM lattice calculations (e.g., plane-wave density-functional theory), which exhibit poor scalability and limited parallelizability.
OpenEye introduces a pioneering approach to optimize crystal structures using QM energy models that are highly parallelizable and scalable across hundreds of thousands of processors. Throughout the CSP calculations, we developed innovative methods to parallelize the problem effectively. With this approach, we can complete CSP for drug-like molecules within a few days of wall clock time, enabling comprehensive exploration for optimizing drug formulation ideas, within your project deadlines.
Overcome the polymorph risks in your drug development process. You provide us with a 2D structure of the API, we provide you with the thermodynamic stability rankings of crystal polymorphs. Discover how OpenEye’s CSP services can assist you by speaking with an expert today.
Webinar: Own Your Own Target with Target X
Webinar: Modular Molecular Modeling
Webinar: ML-Enabled integration of affinity prediction and lead discovery: 3D-QSAR
Webinar: Novel Hits from Beyond the Known: ROCS X
Webinar: Exploring the Uncharted: Discovery at Trillion-Scale with ROCS X
Resources
View Our Recent Webinars
Upcoming Webinar
Webinar: Target X: An Unobstructed View of Pockets
On Demand Webinars
Webinar: Own Your Own Target with Target X



