OpenEye Lead Optimization Solutions
Designed by scientists, for scientists. OpenEye’s fast, robust, and accurate lead optimization tools empower your people looking to design potent and selective molecules. Accessible on the cloud or on your local machines, we give you ultimate flexibility.
Trusted Science.
Shape Alignment
Optimize your lead with rigorous and accurate molecular shape alignment in absence of protein structure. (ROCS®)
Affinity Optimization
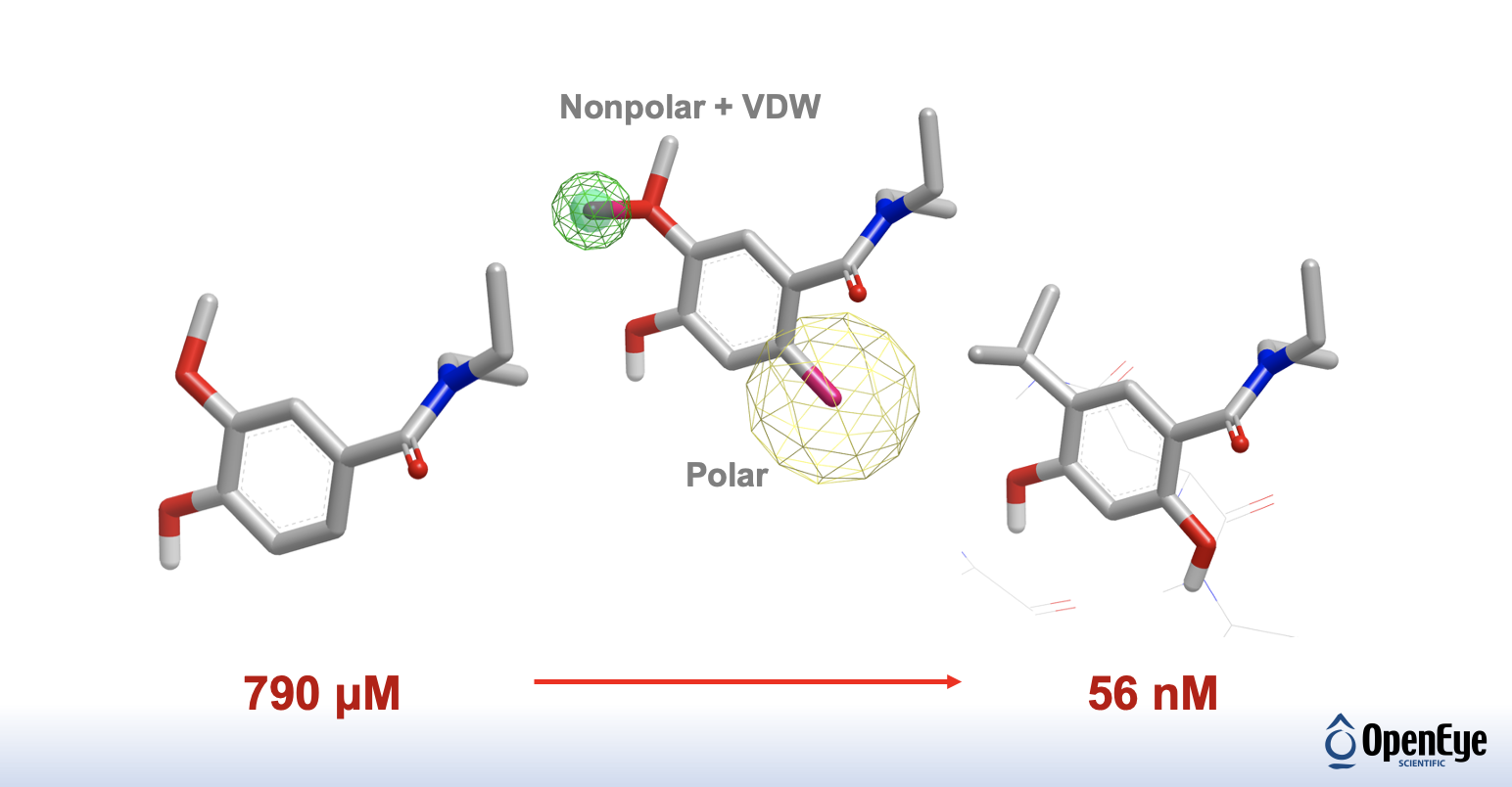
Assess water energetics at specific points along with the protein binding site and explore potential molecular-scaffold manipulations to improve binding affinity. (GamePlan).
Pose Stability
Gain a deep understanding of pose stability by assessing both enthalpic and entropic contributions. (FreeForm).
Pose Prediction
Narrow down which leads are most worthy of your pursuit with fast and accurate pose prediction (POSIT). For flexible protein-targtes, use Induced-Fit Posing to handle side chains flexibility.
pKA Assessment
Get insight into how protonation states may impact your candidates’ physiochemical properties (pKA Prospector).
Delivered the Way You Need.
Webinar: Own Your Own Target with Target X
Webinar: Modular Molecular Modeling
Webinar: ML-Enabled integration of affinity prediction and lead discovery: 3D-QSAR
Webinar: Novel Hits from Beyond the Known: ROCS X
Webinar: Exploring the Uncharted: Discovery at Trillion-Scale with ROCS X
Resources
View Our Recent Webinars
Upcoming Webinar
Webinar: Target X: An Unobstructed View of Pockets
Blog Post
Webinar: Own Your Own Target with Target X


